
3D Bioprinting for Organ Regeneration Market Size, Share | CAGR 16.8%
Global 3D Bioprinting for Organ Regeneration Market Size, Share & Analysis Report By Organ Type (Heart, Liver, Kidney, Lung, Skin, Bone, Ear, Eye, Others), Technology (Extrusion-Based, Laser-Assisted, Inkjet, Stereolithography, Magnetic Levitation, Laser-Based, Others), Material (Bioinks, Polymers, Hydrogels, Ceramics, Metals, Biomaterials, Others), Application, End User, Region & Key Players – Trends & Forecast 2025–2034
Report Overview
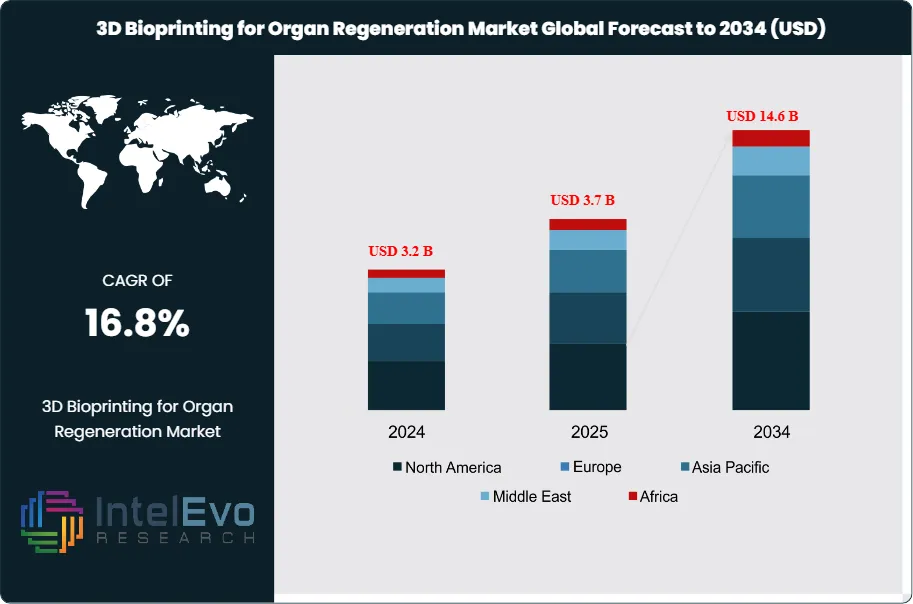
The 3D Bioprinting for Organ Regeneration Market is projected to reach approximately USD 14.6 billion by 2034, rising from USD 3.2 billion in 2024, growing at a CAGR of 16.8% during the forecast period from 2025 to 2034. This strong growth trajectory is driven by rapid advances in bioprinting technologies, stem cell research, and the rising demand for personalized regenerative therapies. Increasing investments in organ tissue engineering and collaboration between biotech firms and healthcare institutions are creating new pathways for life-saving innovations. As 3D bioprinting moves closer to clinical applications, the market is expected to revolutionize transplant medicine and precision healthcare worldwide.
Get More Information about this report -
Request Free Sample ReportThis growth is driven by advancements in bioprinting technologies, rising demand for organ transplantation, and increasing investments in regenerative medicine. Furthermore, supportive government initiatives and collaborations between biotech firms and research institutions are expected to accelerate market adoption over the coming decade.
The 3D Bioprinting for Organ Regeneration Market is the next step in the development of functional tissues and organs using advanced 3D printing technologies. It involves bioinks material, which is composed of living cells along with biomaterials layered to form complex biological structures. These bioprinted tissues may become the solution to some of the world's significant problems such as scarcity of donor organs, rejection of organs, and high costs of transplantation. Third, 3D bioprinting is applied in research, drug testing and regenerative medicine, to replace the services of this element in the healthcare industry.
The market is classified by organ type, technology, material, application, and end-users. The highest-demand organs in bioprinting include the heart, liver, kidney, and skin due to transplants and their complex biological mechanisms. Technologies involve extrusion-based, laser-assisted, and inkjet printing in enabling precise tissue creation. In terms of materials, a bioink, which contains living cells, forms an essential component of bioprinting functional tissues. Applications involve transplantation, disease modeling, drug testing, and medical training.
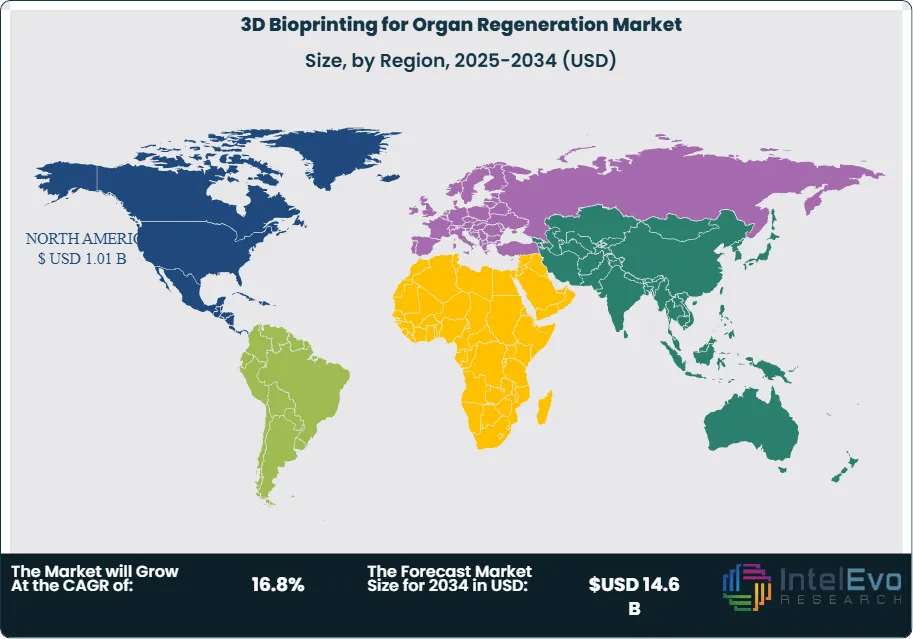
North America holds the highest position in the market, largely because of advanced healthcare infrastructure, high R&D expenditures, and companies that are strong stakeholders in the value chain, such as Organovo and CELLINK. The region is also fueled by the organ shortage and use of regenerative medicine technology. The Asia-Pacific region is expanding rapidly due to increased investment in the healthcare sector, amicable government policies, and a high population with increasing demands for healthcare. China, Japan, and South Korea, individually, lead the way in investing in bioprinting technologies to improve healthcare-related outcomes and meet the health needs of their citizens in organ transplants.

Key Takeaways
- Market Growth: The 3D bioprinting for organ regeneration market is expected to reach USD 14.6 billion by 2034, growing at a robust CAGR of 16.8%, indicating strong market expansion.
- Organ Type Dominance: The liver and heart dominate due to the high demand for transplants and the complexity of their biological functions. Liver tissue is particularly important for drug toxicity testing, while the heart is a critical area of focus for conditions such as cardiovascular disease. Companies like Organovo and BIOLIFE4D are leading the charge in developing liver and heart tissues, making these the most prioritized organ types in bioprinting.
- Technology Preference: Extrusion-based bioprinting is the most widely used technology due to its versatility and ability to print complex structures at high resolutions. It allows for precise layering of bioinks and cells, making it suitable for creating functional tissues. Additionally, it’s cost-effective and scalable, making it the go-to technology for both research and clinical applications.
- Material Dominance: Bioinks, made from living cells, are essential for creating functional tissues that mimic human organs. The success of 3D bioprinting largely depends on the compatibility and functionality of these bioinks, which provide the foundation for tissue growth and regeneration. With ongoing innovations in bioink formulations, this segment remains critical for the overall success of organ regeneration.
- Application Dominance: Transplantation dominates as the primary application due to the increasing global demand for organ transplants and the shortage of donors. Bioprinting aims to address this gap by creating functional organs that can eventually be transplanted into patients. Although still in development, transplantation offers the most direct and impactful use case for 3D bioprinting technologies.
- End-user Preference: Research institutes and laboratories dominate this segment as 3D bioprinting is still largely in the experimental and development phase. These organizations are driving innovation through research into tissue engineering, disease modeling, and drug testing, which are critical precursors to clinical applications in hospitals and pharmaceutical companies.
- Driver: Innovations like extrusion-based printing, laser-assisted methods, and stereolithography enhance bioprinting by enabling precise cell deposition and improved bioinks, facilitating complex tissue modeling for research and clinical applications.
- Restraint: The high costs of bioprinters, bioinks, and specialized expertise hinder 3D bioprinting adoption, especially in smaller institutions and developing regions, limiting access to organ regeneration technologies and delaying clinical use.
- Opportunity: 3D bioprinting enables the creation of patient-specific tissues and organs, reducing immune rejection risks. This personalization leads to tailored treatments, drug testing, and breakthroughs in regenerative medicine and targeted therapies.
- Trend: AI and machine learning enhance 3D bioprinting by optimizing processes, analyzing data on cell behavior and tissue growth, enabling real-time adjustments, predicting tissue responses, and improving organ regeneration and personalized medicine.
- Regional Analysis: North America and Asia-Pacific are projected to be the regions with the highest market share and growth potential for 3D bioprinting for organ regeneration, driven by advanced healthcare, significant R&D investment, supportive regulations, and rising organ transplant demands, and driving adoption of bioprinting solutions.
Organ Type Analysis:
The liver and heart are the most critical parts in 3D bioprinting, because of their high demand rates in transplantation and due to their complexity in the biological process. Liver tissue is found to be crucial in the testing of drug toxicity, where researchers will be able to assess the potential effects that drugs would have on human-like systems. The heart is at the center of cardiovascular disease, which contributes largely to death rates across the globe. A few companies, like Organovo and BIOLIFE4D, are very enthusiastically working towards the direction of bioprinting liver and heart tissues, giving preference to these organs for research and clinical applications and focusing on meeting the need for organ transplant at the earliest.
Technology Analysis:
Extrusion-based 3D bioprinting is the most popular technology being used mainly because of its flexibility and its potential to create complex structures with excellent resolution. This technique allows the bioinks and the living cells to be deposited at different exact depths, hence allowing the creation of functional tissues that closely resemble the natures. Its cost-effectiveness and scalability are extremely important in both applied research and clinical setups. In addition, extrusion-based bioprinting can be modified to use various types of materials to give rise to many tissue types. Its being on the cutting edge of technology grants this to be the first choice for researchers and practitioners working on advancing the regeneration of organs.
Material Analysis:
Biological materials, which consist of living cells in the main composition, are responsible for the effectiveness of 3D bioprinting as well as functional tissues with features comparable to human organs. In total, it is the performance and compatibility of these bioinks that determine bioprinting efficiency because they establish an environment in which tissue growth and regeneration are possible. Advances in bioink formulation incorporate improved biocompatibility and designed properties for enhanced functionality. Such formulations are one among the key success factors for efforts into organ regeneration. Bioinks are getting more complex with research; it continues to consolidate their importance in the bioprinting landscape.
Application Analysis:
The most relevant application of 3D bioprinting is organ transplantation because of the overall increased global need for transplantation and the on-going organ donor shortage. Possibly, 3D bioprinting could solve this problem by creating organs that could function, and eventually, be implanted in patients and serve as a cure for such life-threatening conditions. Although still in its development phase, the actual usage case is to develop functional organs through bioprinting. Scientists are actively studying techniques to make bioprinted organs functional and compatible, thus widening what may ultimately be possible with organ transplants.
End-user Analysis:
Research centers and laboratories take the maximum portion of the 3D bioprinting market, simply because the technology is in its experimental stage and has been devised for development. These organizations spur further innovations through significant opportunities in research work related to tissue engineering, disease models, and drug testing. Their findings represent crucial precursors to eventual clinical applications, and therefore serve to empower hospitals and pharmaceutical corporations in their use of bioprinting technologies. Bioprinted tissues and organs will lead to advanced research on organ regeneration, personalized medicine, and therapeutic solutions for the future. They shall pave the way for the future of patient care in many medical disciplines.
Region Analysis:
North America Leads With 35% Market Share In 3D bioprinting for organ regeneration market. North America leads the market due to the region's advanced healthcare infrastructure, substantial investment in research and development, and the presence of key market players like Organovo and CELLINK. The U.S. also has well-established regulatory frameworks supporting innovation in biotechnology and organ regeneration. Additionally, North America’s robust funding ecosystem, driven by both public institutions (like the NIH) and private investments, accelerates 3D bioprinting advancements. This region's demand is further fueled by the rising number of organ transplants and the shortage of organ donors, prompting increased adoption of 3D bioprinting solutions.
Asia-Pacific is witnessing the fastest growth, driven by increasing healthcare investments, growing awareness about regenerative medicine, and rapid technological adoption in countries like China, Japan, and South Korea. These nations are heavily investing in biotechnology research, supported by government initiatives to enhance healthcare outcomes. Additionally, the region benefits from a growing population, increasing prevalence of chronic diseases, and a rise in research collaborations between academic institutions and biotech firms, fostering innovation in 3D bioprinting technologies. The expanding biotech industry and supportive regulatory environment further propel growth in this region.
Get More Information about this report -
Request Free Sample Report
Key Market Segment
Organ Type:
- Heart
- Liver
- Kidney
- Lung
- Skin
- Bone
- Ear
- Eye
- Others
By Component
- Bioprinters
- Bioinks
- Software & Services
By Material
- Biological Materials (Bioinks)
- Synthetic Polymers
- Hydrogels
- Ceramics
- Metals
- Biomaterials
- Others
By Technology
- Inkjet 3D Bioprinting
- Laser-assisted Bioprinting
- Extrusion-based Bioprinting
- Magnetic Bioprinting
- Stereolithography
- Laser Based
- Others
By End User
- Research & Academic Institutions
- Pharmaceutical & Biotechnology Companies
- Hospitals & Transplant Centers
- Contract Research Organizations (CROs)
- Others
By Application
- Organ Regeneration (Liver, Kidney, Heart, Lungs, Others)
- Tissue Engineering
- Drug Testing & Research
- Cosmetic & Clinical Applications
- Others
Region:
- North America
- Latin America
- East Asia And Pacific
- Sea And South Asia
- Eastern Europe
- Western Europe
- Middle East & Africa
| Report Attribute | Details |
| Market size (2025) | USD 3.2 B |
| Forecast Revenue (2034) | USD 14.6 B |
| CAGR (2025-2034) | 16.8% |
| Historical data | 2020-2023 |
| Base Year For Estimation | 2024 |
| Forecast Period | 2025-2034 |
| Report coverage | Revenue Forecast, Competitive Landscape, Market Dynamics, Growth Factors, Trends and Recent Developments |
| Segments covered | By Organ Type (Heart, Liver, Kidney, Lung, Skin, Bone, Ear, Eye, Others), Technology (Extrusion-Based 3D Printing, Laser-Assisted Printing, Inkjet Printing, Stereolithography, Magnetic Levitation, Laser Based, Others), Material (Biological Materials (Bioinks), Synthetic Polymers, Hydrogels, Ceramics, Metals, Biomaterials, Others), Application (Transplantation, Research & Drug Testing, Disease Modeling, Regenerative Medicine, Medical Training & Education, Others), End User (Hospitals & Clinics, Research Institutes & Laboratories, Pharmaceutical Companies, Academic Institutions, Others), Region |
| Research Methodology |
|
| Regional scope |
|
| Competitive Landscape | Stratasys Ltd., Materialise N.V., BIOLIFE4D, Organovo Holding, Inc., CELLINK , Aspect Biosystems, Cyfuse Biomedical, TeVido Biodevices, Advanced Solutions, Inc., TRS – Tissue Regeneration Systems, Nscrypt, Inc, EnvisionTEC, MedPrin, Nano3D Sciences, Rokit, Cellbricks, REGEMAT 3D, Allevi, Poietis, T&R BIOFAB |
| Customization Scope | Customization for segments, region/country-level will be provided. Moreover, additional customization can be done based on the requirements. |
| Pricing and Purchase Options | Avail customized purchase options to meet your exact research needs. We have three licenses to opt for: Single User License, Multi-User License (Up to 5 Users), Corporate Use License (Unlimited User and Printable PDF). |
Frequently Asked Questions
How big is the 3D Bioprinting for Organ Regeneration Market?
3D Bioprinting for Organ Regeneration Market to reach USD 14.6 Bn by 2034, driven by tech advances, rising transplants, and regenerative medicine growth.
Who are the major players in the 3D Bioprinting for Organ Regeneration Market?
Stratasys Ltd., Materialise N.V., BIOLIFE4D, Organovo Holding, Inc., CELLINK , Aspect Biosystems, Cyfuse Biomedical, TeVido Biodevices, Advanced Solutions, Inc., TRS – Tissue Regeneration Systems, Nscrypt, Inc, EnvisionTEC, MedPrin, Nano3D Sciences, Rokit, Cellbricks, REGEMAT 3D, Allevi, Poietis, T&R BIOFAB
Which segments covered the 3D Bioprinting for Organ Regeneration Market?
By Organ Type (Heart, Liver, Kidney, Lung, Skin, Bone, Ear, Eye, Others), Technology (Extrusion-Based 3D Printing, Laser-Assisted Printing, Inkjet Printing, Stereolithography, Magnetic Levitation, Laser Based, Others), Material (Biological Materials (Bioinks), Synthetic Polymers, Hydrogels, Ceramics, Metals, Biomaterials, Others), Application (Transplantation, Research & Drug Testing, Disease Modeling, Regenerative Medicine, Medical Training & Education, Others), End User (Hospitals & Clinics, Research Institutes & Laboratories, Pharmaceutical Companies, Academic Institutions, Others), Region
How can this market research report help my business make strategic decisions?
Our market research reports provide actionable intelligence, including verified market size data, CAGR projections, competitive benchmarking, and segment-level opportunity analysis. These insights support strategic planning, investment decisions, product development, and market entry strategies for enterprises and startups alike.
How frequently is the data updated?
We continuously monitor industry developments and update our reports to reflect regulatory changes, technological advancements, and macroeconomic shifts. Updated editions ensure you receive the latest market intelligence.
Select Licence Type
Connect with our sales team
3D Bioprinting for Organ Regeneration Market
Published Date : 30 Oct 2024 | Formats :Why IntelEvoResearch
100%
Customer
Satisfaction
24x7+
Availability - we are always
there when you need us
200+
Fortune 50 Companies trust
IntelEvoResearch
80%
of our reports are exclusive
and first in the industry
100%
more data
and analysis
1000+
reports published
till date







