
Global Antisense Oligonucleotide Market Forecast 2034 | CAGR 12.1%
Global Antisense Oligonucleotide Market Size, Share, Growth & Industry Analysis By Mechanism of Action (RNase H-Mediated Gapmers, Splice-Modulating Mixmers, Translation-Blocking, Others), By Therapeutic Area (Neurological Disorders, Rare Genetic Diseases, Cardiovascular & Metabolic Diseases, Oncology, Infectious Diseases, Ophthalmology), By Route of Administration (Subcutaneous, Intrathecal, Intravenous, Intravitreal), By End User (Hospitals, Clinics, Pharmacies, Research Institutes) Industry Trends & Forecast 2026–2034
Report Overview
| Market Size (2025) | Forecast Value (2034) | CAGR (2026–2034) | Largest Region (2025) |
| USD 4.6 Billion | USD 12.8 Billion | 12.1% | North America, 52.4% |
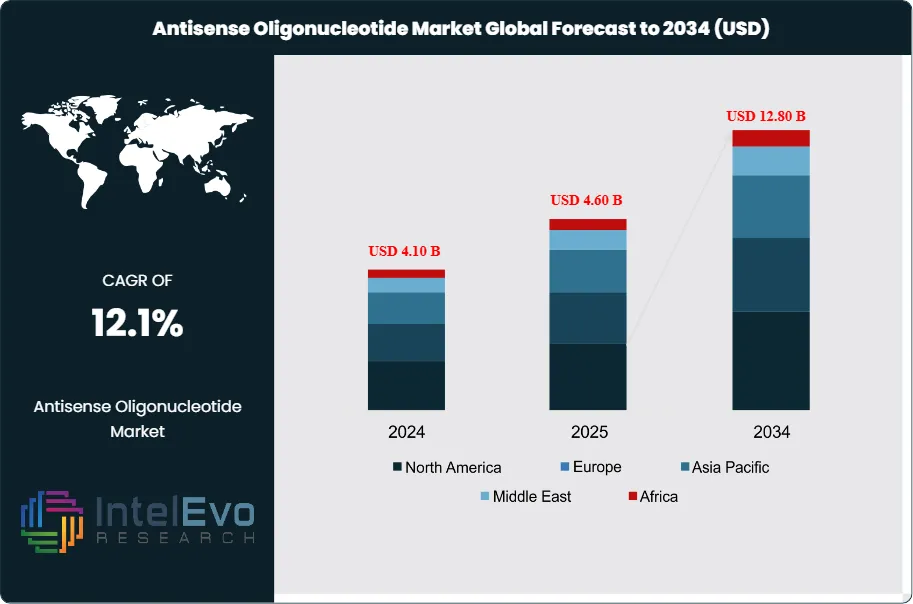
The Antisense Oligonucleotide Market was valued at approximately USD 4.10 Billion in 2024 and reached USD 4.60 Billion in 2025. The market is projected to grow to USD 12.80 Billion by 2034, expanding at a CAGR of 12.1% during the forecast period from 2026 to 2034. This represents an absolute dollar opportunity of USD 8.2 Billion over the analysis period. Antisense oligonucleotides (ASOs) have established a validated therapeutic modality for treating genetic diseases by modulating gene expression through sequence-specific RNA binding. These synthetic single-stranded nucleic acid sequences, typically 15-25 nucleotides in length, bind complementary mRNA targets through Watson-Crick base pairing to achieve gene knockdown via RNase H-mediated degradation or splice modulation through steric blocking mechanisms.
Get More Information about this report -
Request Free Sample ReportThe antisense oligonucleotide market has matured significantly following FDA approvals of multiple ASO therapies between 2016 and 2025. Ionis Pharmaceuticals has pioneered this modality with three commercially approved products: nusinersen (Spinraza) for spinal muscular atrophy, inotersen (Tegsedi) for hereditary transthyretin amyloidosis, and tofersen (Qalsody) for SOD1-ALS. Combined commercial revenue from approved ASO therapies exceeded USD 2.8 Billion in 2025. The clinical pipeline contains over 180 ASO candidates in active development across neurological, cardiovascular, metabolic, and rare disease therapeutic areas.
Chemistry advances have transformed ASO therapeutic profiles over the past decade. Generation 2+ modifications including constrained ethyl (cEt) and 2'-O-methoxyethyl (2'-MOE) substitutions have improved binding affinity, nuclease resistance, and pharmacokinetic properties. Ligand-conjugated antisense (LICA) technology using GalNAc targeting has enabled subcutaneous dosing with extended dosing intervals for hepatic indications. ICH guidelines (M4E, S6R1) provide standardized regulatory requirements for oligonucleotide development. North America dominates with 52.4% market share driven by Ionis Pharmaceuticals' leadership, favorable FDA pathways including Breakthrough Therapy and Accelerated Approval designations, and robust specialty pharmacy infrastructure supporting high-cost genetic medicines. Asia Pacific represents the fastest-growing region at 14.6% CAGR as contract manufacturing expands and regulatory harmonization with FDA/EMA standards accelerates market access.

Key Takeaways
- Market Growth: The antisense oligonucleotide market grows from USD 4.6 Billion in 2025 to USD 12.8 Billion by 2034, registering a CAGR of 12.1% across the forecast period 2026–2034.
- Segment Dominance: RNase H-mediated ASOs lead the market by mechanism at 58.6% share in 2025, supported by validated clinical programs and extensive manufacturing experience across multiple therapeutic areas.
- Segment Dominance: Neurological disorders represent the largest therapeutic application at 44.8% market share in 2025, anchored by nusinersen (Spinraza) revenue exceeding USD 1.8 Billion annually.
- Driver: Expanding rare disease applications drive adoption with over 7,000 identified genetic disorders and only 5% having approved treatments. Orphan Drug Act incentives have increased ASO orphan designations 6-fold since 2015.
- Restraint: High therapy costs limit market penetration with annual treatment expenses ranging from USD 300,000 to USD 750,000 per patient. Payer prior authorization delays treatment initiation by an average of 38 days.
- Opportunity: Cardiovascular disease expansion presents a USD 2.8 Billion opportunity by 2034. Phase III programs targeting APOC3, Lp(a), and ANGPTL3 have demonstrated 60-80% target reduction in registrational trials.
- Trend: LICA (Ligand-Conjugated Antisense) technology has achieved 72% adoption rate among liver-targeted ASO programs in development, enabling monthly to quarterly subcutaneous dosing regimens.
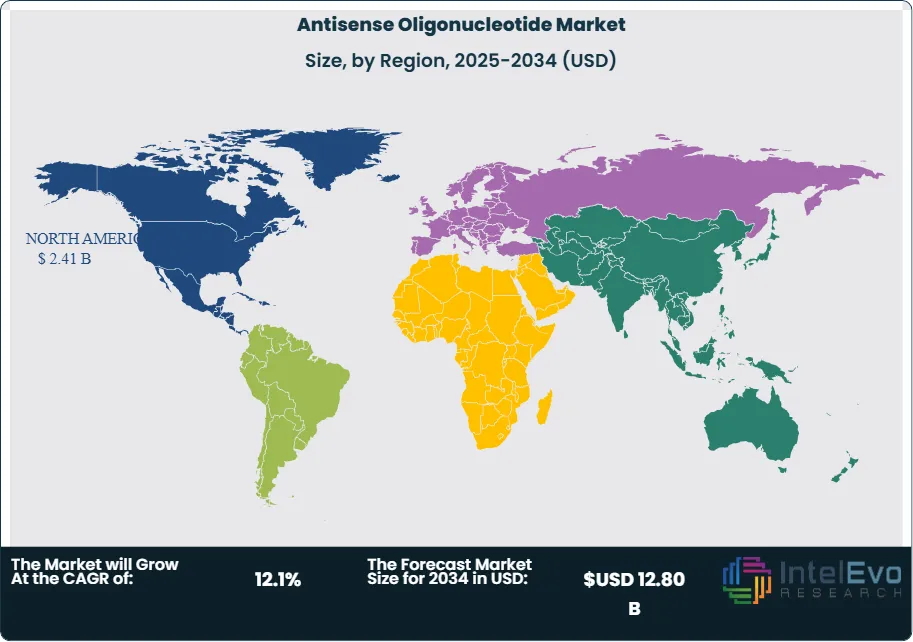
- Regional Analysis: North America commands 52.4% market share valued at USD 2.41 Billion in 2025, driven by Ionis Pharmaceuticals' commercial leadership and favorable FDA regulatory pathways for genetic medicines.
Competitive Landscape Overview
The antisense oligonucleotide market exhibits high consolidation with the top four companies controlling approximately 72% of global revenue in 2025. Ionis Pharmaceuticals dominates with over 48% market share through proprietary ASO chemistry platforms and three FDA-approved products. Competition centers on therapeutic area expansion, chemistry differentiation, and delivery technology advancement. Strategic partnerships between Ionis and large pharmaceutical companies including Biogen, Roche, AstraZeneca, and Novartis have defined competitive dynamics, with cumulative deal values exceeding USD 8 Billion since 2020. Recent competitive intensity has increased as second-generation players including Sarepta Therapeutics and Wave Life Sciences advance differentiated ASO platforms toward late-stage clinical development.
Competitive Landscape Matrix
| Company | HQ | Position | Key Product | Geo Strength | Recent Strategic Move |
| Ionis Pharmaceuticals | US | Leader | Spinraza (nusinersen) | North America | FDA approval of eplontersen for ATTR cardiomyopathy (Dec 2024) |
| Sarepta Therapeutics | US | Leader | Exondys 51 (eteplirsen) | North America | Initiated Phase III for SRP-5051 peptide-PMO (Jan 2026) |
| Biogen | US | Leader | Spinraza (commercial) | Global | Phase III initiation for ION582 in Angelman syndrome (Feb 2026) |
| Wave Life Sciences | US | Challenger | WVE-N531 (DMD exon 53) | North America | Positive Phase II data in DMD with 28% dystrophin (Mar 2025) |
| Roche | Switzerland | Challenger | Tominersen (HD, paused) | Europe | Resumed development of HD ASO with modified dosing (Aug 2025) |
| Novartis | Switzerland | Challenger | Pelacarsen (Lp(a)) | Global | Expanded Ionis partnership with USD 500M milestones (Sep 2025) |
| GSK | UK | Challenger | Bepirovirsen (HBV) | Europe | Phase III completion for chronic HBV functional cure (Jul 2025) |
| AstraZeneca | UK | Niche Player | Eplontersen (CV) | Global | Launched commercial partnership with Ionis for eplontersen (Jan 2025) |
| Stoke Therapeutics | US | Niche Player | STK-001 (Dravet) | North America | Advanced Phase III for Dravet syndrome ASO (Apr 2025) |
| Nippon Shinyaku | Japan | Niche Player | Viltolarsen (DMD) | Asia Pacific | Expanded viltolarsen into 15 additional markets (Jun 2025) |
By Mechanism of Action
RNase H-mediated antisense oligonucleotides dominate the market by mechanism at 58.6% share valued at USD 2.70 Billion in 2025. This mechanism employs ASOs with DNA-like central regions (gapmers) that recruit cellular RNase H enzyme to cleave the target mRNA, achieving potent and durable gene knockdown. Ionis Pharmaceuticals has commercialized multiple RNase H ASOs including inotersen (Tegsedi) for hATTR and tofersen (Qalsody) for SOD1-ALS. The gapmer design benefits from extensive clinical validation across over 120 clinical programs and well-characterized safety profiles. Manufacturing processes for RNase H ASOs have achieved cost optimization through solid-phase synthesis improvements and yield enhancements exceeding 85% at commercial scale.
Splice-modulating antisense oligonucleotides represent 32.8% market share at USD 1.51 Billion in 2025. These fully modified ASOs (mixmers) sterically block splicing machinery without recruiting RNase H, enabling exon inclusion or exclusion to restore functional protein expression. Nusinersen (Spinraza) for spinal muscular atrophy established the splice-modulation market, generating annual revenue exceeding USD 1.8 Billion globally. Sarepta Therapeutics has commercialized four exon-skipping ASOs for Duchenne muscular dystrophy: eteplirsen (Exondys 51), golodirsen (Vyondys 53), casimersen (Amondys 45), and viltolarsen (Viltepso). The splice-modulation approach enables therapeutic intervention in diseases where complete gene knockdown would be harmful but restoring partial function provides clinical benefit.
Translation-blocking antisense oligonucleotides account for 8.6% market share at USD 0.39 Billion in 2025. These ASOs target the 5' untranslated region or start codon area of mRNA to prevent ribosome assembly and translation initiation without inducing mRNA degradation. The mechanism suits applications where transient protein reduction is desired or where target mRNA stability provides pharmacodynamic advantages. Clinical programs using translation blocking approaches have demonstrated utility in oncology settings where cancer cells may develop resistance to complete target elimination. Other mechanisms collectively represent approximately 5% of the market.
By Therapeutic Area
Neurological disorders lead therapeutic applications for the antisense oligonucleotide market at 44.8% share valued at USD 2.06 Billion in 2025. Spinal muscular atrophy treatment with nusinersen (Spinraza) anchors this segment with global revenue exceeding USD 1.8 Billion annually. Intrathecal delivery enables direct CNS penetration, bypassing blood-brain barrier limitations that constrain systemically administered therapies. The FDA approved tofersen (Qalsody) for SOD1-ALS in 2023, expanding the neurological ASO portfolio. Clinical programs targeting Huntington's disease, Alzheimer's disease, Parkinson's disease, and Alexander disease are advancing through Phase II and Phase III development. The segment benefits from established specialty neurology centers capable of intrathecal administration and monitoring.
Rare genetic diseases represent 26.4% market share at USD 1.21 Billion in 2025. Hereditary transthyretin amyloidosis treatment with inotersen (Tegsedi) demonstrates ASO efficacy in systemic genetic conditions. Over 7,000 identified rare diseases affect approximately 400 million people globally, with 80% having genetic etiology addressable through ASO intervention. Orphan Drug Act economic incentives including seven-year market exclusivity, FDA fee waivers, and tax credits have accelerated rare disease ASO development. The pipeline contains over 60 ASO candidates targeting rare metabolic, hematologic, and musculoskeletal conditions. Patient identification challenges and small population sizes require specialized commercial models including patient registries and genetic testing partnerships.
Cardiovascular and metabolic diseases account for 18.2% market share at USD 0.84 Billion in 2025. Ionis and partners have advanced multiple cardiovascular ASO programs targeting APOC3, lipoprotein(a), and ANGPTL3 for dyslipidemia indications. Olezarsen targeting APOC3 received FDA approval in 2025 for familial chylomicronemia syndrome. Pelacarsen targeting lipoprotein(a) has completed Phase III trials with cardiovascular outcomes data. GalNAc-conjugated ASOs enable subcutaneous self-administration with monthly to quarterly dosing, improving convenience over intravenous alternatives. The cardiovascular segment positions ASOs for chronic disease populations significantly larger than traditional rare disease markets.
Oncology represents 7.4% market share at USD 0.34 Billion in 2025, with strong growth potential. ASO approaches in cancer target oncogenes, tumor suppressors, and immunomodulatory pathways. Clinical programs address KRAS, STAT3, BCL-2, and other validated cancer targets. Delivery to solid tumors remains challenging, driving development of lipid nanoparticle formulations and antibody-ASO conjugates for tumor-selective delivery. Combination approaches with checkpoint inhibitors and targeted therapies expand ASO utility in oncology settings. Infectious diseases and ophthalmology collectively represent the remaining 3.2% market share.
By Route of Administration
Subcutaneous injection leads the antisense oligonucleotide market by administration route at 48.2% share valued at USD 2.22 Billion in 2025. GalNAc conjugation and LICA technology have transformed ASO delivery by enabling hepatocyte-specific uptake through asialoglycoprotein receptor binding. Subcutaneous administration supports self-injection at home or clinic-based dosing with intervals ranging from weekly to monthly depending on the specific ASO pharmacokinetics. The route dominates cardiovascular and metabolic ASO programs where liver-expressed targets drive disease pathology. Patient convenience studies demonstrate strong preference for subcutaneous over intravenous alternatives when efficacy is equivalent.
Intrathecal administration represents 38.6% market share at USD 1.78 Billion in 2025. Direct CNS delivery bypasses blood-brain barrier limitations for neurological indications. Nusinersen (Spinraza) established intrathecal ASO administration through lumbar puncture every four months following loading doses. Tofersen (Qalsody) for SOD1-ALS employs similar intrathecal delivery schedules. The route requires specialized healthcare facilities and trained personnel for lumbar puncture procedures. Novel intrathecal pump systems and direct CNS injection devices are under development to reduce procedural burden for chronic neurological conditions requiring repeated administration.
Intravenous infusion accounts for 9.8% market share at USD 0.45 Billion in 2025. Systemic IV delivery enables broad tissue distribution for indications requiring exposure beyond liver or CNS. Early-generation ASOs without targeting ligands required IV administration to achieve therapeutic concentrations. The route persists for oncology applications and certain rare disease indications. Intravenous ASOs require healthcare facility administration and monitoring during infusion. Intravitreal and other routes represent the remaining 3.4% share, primarily serving ophthalmology applications including retinal disorders.
By End User
Hospitals dominate the antisense oligonucleotide market by end user at 52.4% share valued at USD 2.41 Billion in 2025. Academic medical centers and specialty hospitals provide infrastructure for intrathecal administration, infusion services, and complex patient monitoring. Neurology departments at tertiary care centers have concentrated expertise in SMA diagnosis and nusinersen treatment initiation. Hospital pharmacy formulary decisions drive institutional access, with value-based contracts linking reimbursement to clinical outcomes becoming standard for high-cost ASO therapies. Integrated delivery networks leverage purchasing volume to negotiate pricing with manufacturers.
Specialty pharmacies represent 34.2% market share at USD 1.57 Billion in 2025. Limited distribution networks restrict ASO access to designated specialty pharmacy partners ensuring proper handling, patient education, and adherence monitoring. Hub service models coordinate among prescribers, payers, and pharmacies to facilitate prior authorization, benefits investigation, and treatment initiation. Cold chain management requirements for certain ASO formulations necessitate specialized distribution capabilities. Patient support programs operated through specialty pharmacies have improved treatment persistence rates to 78% at 12 months for chronic ASO therapies.
Clinics and ambulatory care centers account for 10.2% market share at USD 0.47 Billion in 2025. Subcutaneous ASO therapies with extended dosing intervals enable clinic-based treatment without hospital infrastructure. Cardiologist offices increasingly administer GalNAc-conjugated ASOs for cardiovascular indications. The segment grows as convenience-optimized ASOs expand beyond rare disease into chronic disease populations. Research institutions represent the remaining 3.2% share, supporting clinical trials and academic translational research programs.
Regional Analysis
North America Antisense Oligonucleotide Market
North America commands 52.4% of the global antisense oligonucleotide market, valued at USD 2.41 Billion in 2025. The United States represents 94% of regional revenue at USD 2.27 Billion, establishing clear dominance through the concentration of ASO innovator companies and favorable regulatory pathways. Ionis Pharmaceuticals (Carlsbad, California) has built the largest ASO pipeline globally with over 45 clinical programs and three FDA-approved products. Sarepta Therapeutics (Cambridge, Massachusetts) complements with exon-skipping ASOs for Duchenne muscular dystrophy. Biogen's commercial partnership with Ionis drives nusinersen distribution across neurology specialty centers.
FDA regulatory frameworks provide multiple expedited pathways accelerating ASO development timelines. Breakthrough Therapy Designation has been granted to over 18 ASO programs since 2012, enabling intensive FDA guidance and rolling review. Accelerated Approval based on surrogate biomarker endpoints facilitated early market access for tofersen and exon-skipping DMD therapies pending confirmatory trials. The Inflation Reduction Act's orphan drug exemption from Medicare price negotiation preserves economic incentives for rare disease ASO development through 2028. Specialty pharmacy infrastructure supporting high-cost therapies and established patient access programs facilitate commercial uptake. Canada contributes USD 140 million in 2025 market value with regulatory alignment through Health Canada's Notice of Compliance process.
Europe Antisense Oligonucleotide Market
Europe accounts for 26.8% market share valued at USD 1.23 Billion in 2025 for antisense oligonucleotides. Germany leads regional adoption at USD 340 million (27.6% of Europe), driven by SHI (Statutory Health Insurance) coverage decisions and strong pharmaceutical industry presence. The UK market at USD 265 million benefits from NHS England's managed access agreements providing early patient access while generating real-world evidence for health technology assessment. France at USD 225 million maintains centralized HTA evaluation through Haute Autorite de Sante determining pricing and reimbursement eligibility.
European Medicines Agency (EMA) centralized marketing authorization provides single approval across 27 EU member states plus EEA countries. Conditional Marketing Authorization enables earlier access for ASO therapies addressing unmet medical needs while confirmatory data collection continues. The Committee for Medicinal Products for Human Use (CHMP) has established assessment expertise for oligonucleotide modalities through multiple approval procedures. Italy and Spain collectively represent USD 280 million in 2025 market value with national pricing negotiations following EMA approval. Contract manufacturing capacity in Ireland and Switzerland supports global ASO supply chains with specialized solid-phase synthesis capabilities.
Asia Pacific Antisense Oligonucleotide Market
Asia Pacific represents 14.6% market share valued at USD 0.67 Billion in 2025, positioned as the fastest-growing region at 14.6% CAGR through 2034 for the antisense oligonucleotide market. Japan contributes USD 285 million (42.5% of regional revenue) with advanced regulatory frameworks and high willingness-to-pay for genetic medicines. PMDA has approved nusinersen and developed assessment pathways for oligonucleotide therapies aligning with ICH guidelines. Japanese pharmaceutical companies including Daiichi Sankyo and Takeda have established ASO development capabilities through licensing agreements with Ionis Pharmaceuticals.
China's antisense oligonucleotide market reaches USD 195 million in 2025 with 16.8% growth projected annually through 2034. NMPA regulatory reforms have accelerated approval timelines with conditional approval pathways available for therapies addressing unmet needs. Domestic companies including Hansoh Pharma are advancing proprietary ASO pipelines targeting Chinese patient populations. Contract manufacturing capacity expansion positions China as an emerging oligonucleotide synthesis hub. South Korea contributes USD 95 million with Samsung Biologics investing in nucleic acid manufacturing capabilities. India at USD 62 million serves primarily as a CDMO destination for Western pharmaceutical companies requiring ASO synthesis capacity. Australia rounds out regional activity with USD 35 million in market value and active clinical trial participation.
Latin America Antisense Oligonucleotide Market
Latin America holds 3.6% market share valued at USD 0.17 Billion in 2025 for antisense oligonucleotides. Brazil dominates regional activity at USD 92 million (54.1% of Latin America), serving as the primary market entry point for specialty pharmaceutical products. ANVISA regulatory pathways incorporate expedited review for rare disease therapies, though infrastructure limitations constrain intrathecal ASO administration to major metropolitan centers. The Brazilian Unified Health System (SUS) has incorporated nusinersen following CONITEC technology assessment, establishing precedent for ASO coverage.
Mexico contributes USD 45 million with proximity to US pharmaceutical supply chains facilitating product access. COFEPRIS regulatory alignment with FDA procedures enables technology transfer and manufacturing partnerships. Argentina at USD 22 million maintains clinical trial activity despite currency challenges affecting commercial market development. Colombia and Chile collectively represent the remainder of regional activity with emerging specialty neurology and genetic medicine capabilities. Regional growth at 10.8% CAGR through 2034 remains constrained by limited specialty care infrastructure and reimbursement frameworks for high-cost genetic medicines.
Middle East & Africa Antisense Oligonucleotide Market
Middle East & Africa accounts for 2.6% market share valued at USD 0.12 Billion in 2025 for the antisense oligonucleotide market. The United Arab Emirates leads regional adoption at USD 38 million, driven by Dubai Healthcare City and Abu Dhabi's advanced healthcare infrastructure. UAE genetic medicine access programs have facilitated nusinersen availability for Emirati citizens with spinal muscular atrophy. Saudi Arabia contributes USD 35 million with Vision 2030 healthcare investments prioritizing specialty center development and pharmaceutical localization.
Israel at USD 26 million benefits from advanced biotechnology research and inclusion in European regulatory reference systems. South Africa represents the primary sub-Saharan market at USD 14 million, with specialty pharmaceutical distribution limited to Johannesburg, Cape Town, and Durban metropolitan areas. High therapy costs relative to regional healthcare budgets and limited specialty neurology infrastructure constrain broader market penetration. Regional growth at 11.4% CAGR through 2034 reflects healthcare modernization investments in Gulf Cooperation Council countries, though access disparities persist across the broader MEA region.
Get More Information about this report -
Request Free Sample ReportMarket Key Segments
By Mechanism of Action
- RNase H-Mediated (Gapmers)
- Splice-Modulating (Mixmers)
- Translation-Blocking
- Others
By Therapeutic Area
- Neurological Disorders
- Rare Genetic Diseases
- Cardiovascular and Metabolic Diseases
- Oncology
- Infectious Diseases
- Ophthalmology
By Route of Administration
- Subcutaneous Injection
- Intrathecal Administration
- Intravenous Infusion
- Intravitreal Injection
- Others
By End User
- Hospitals
- Specialty Pharmacies
- Clinics and Ambulatory Care Centers
- Research Institutions
Regional Analysis and Coverage
- North America
- Latin America
- East Asia And Pacific
- Sea And South Asia
- Eastern Europe
- Western Europe
- Middle East & Africa
| Report Attribute | Details |
| Market size (2025) | USD 4.60 B |
| Forecast Revenue (2034) | USD 12.80 B |
| CAGR (2025-2034) | 12.1% |
| Historical data | 2021-2024 |
| Base Year For Estimation | 2025 |
| Forecast Period | 2026-2034 |
| Report coverage | Revenue Forecast, Competitive Landscape, Market Dynamics, Growth Factors, Trends and Recent Developments |
| Segments covered | By Mechanism of Action, (RNase H-Mediated (Gapmers), Splice-Modulating (Mixmers), Translation-Blocking, Others), By Therapeutic Area, (Neurological Disorders, Rare Genetic Diseases, Cardiovascular and Metabolic Diseases, Oncology, Infectious Diseases, Ophthalmology), By Route of Administration, (Subcutaneous Injection, Intrathecal Administration, Intravenous Infusion, Intravitreal Injection, Others), By End User, (Hospitals, Specialty Pharmacies, Clinics and Ambulatory Care Centers, Research Institutions) |
| Research Methodology |
|
| Regional scope |
|
| Competitive Landscape | IONIS PHARMACEUTICALS, SAREPTA THERAPEUTICS, BIOGEN, WAVE LIFE SCIENCES, ROCHE, NOVARTIS, ASTRAZENECA, GSK, DAIICHI SANKYO, NIPPON SHINYAKU, STOKE THERAPEUTICS, PROQR THERAPEUTICS, DYNACURE, ULTRAGENYX PHARMACEUTICAL, PTC THERAPEUTICS, Others |
| Customization Scope | Customization for segments, region/country-level will be provided. Moreover, additional customization can be done based on the requirements. |
| Pricing and Purchase Options | Avail customized purchase options to meet your exact research needs. We have three licenses to opt for: Single User License, Multi-User License (Up to 5 Users), Corporate Use License (Unlimited User and Printable PDF). |
Frequently Asked Questions
How big is the Antisense Oligonucleotide Market?
Global Antisense oligonucleotide market valued at USD 4.10B in 2024, reaching USD 12.8B by 2034, growing at a CAGR of 12.1% from 2026–2034.
Who are the major players in the Antisense Oligonucleotide Market?
IONIS PHARMACEUTICALS, SAREPTA THERAPEUTICS, BIOGEN, WAVE LIFE SCIENCES, ROCHE, NOVARTIS, ASTRAZENECA, GSK, DAIICHI SANKYO, NIPPON SHINYAKU, STOKE THERAPEUTICS, PROQR THERAPEUTICS, DYNACURE, ULTRAGENYX PHARMACEUTICAL, PTC THERAPEUTICS, Others
Which segments covered the Antisense Oligonucleotide Market?
By Mechanism of Action, (RNase H-Mediated (Gapmers), Splice-Modulating (Mixmers), Translation-Blocking, Others), By Therapeutic Area, (Neurological Disorders, Rare Genetic Diseases, Cardiovascular and Metabolic Diseases, Oncology, Infectious Diseases, Ophthalmology), By Route of Administration, (Subcutaneous Injection, Intrathecal Administration, Intravenous Infusion, Intravitreal Injection, Others), By End User, (Hospitals, Specialty Pharmacies, Clinics and Ambulatory Care Centers, Research Institutions)
How can this market research report help my business make strategic decisions?
Our market research reports provide actionable intelligence, including verified market size data, CAGR projections, competitive benchmarking, and segment-level opportunity analysis. These insights support strategic planning, investment decisions, product development, and market entry strategies for enterprises and startups alike.
How frequently is the data updated?
We continuously monitor industry developments and update our reports to reflect regulatory changes, technological advancements, and macroeconomic shifts. Updated editions ensure you receive the latest market intelligence.
Select Licence Type
Connect with our sales team
Antisense Oligonucleotide Market
Published Date : 10 Apr 2026 | Formats :Why IntelEvoResearch
100%
Customer
Satisfaction
24x7+
Availability - we are always
there when you need us
200+
Fortune 50 Companies trust
IntelEvoResearch
80%
of our reports are exclusive
and first in the industry
100%
more data
and analysis
1000+
reports published
till date







