
Rare Disease Treatment Market 2034 | Gene Therapy, Orphan Drugs & Precision Medicine
Global Rare Disease Treatment Innovation Market Size, Share, Analysis Report By Disease Type(Cancer, Metabolic Diseases, Neurological Conditions, Hematologic Disorders, Infectious Diseases, Musculoskeletal Disorders, Cardiovascular Disorders, Others), Type(Biologics, Non-Biologics), End-User(Specialty Pharmacies, Hospital Pharmacies, Retail Pharmacies), Age Group(Adult, Pediatric), Region and Key Players - Industry Segment Overview, Market Dynamics, Competitive Strategies, Trends and Forecast 2025-2034
Report Overview:
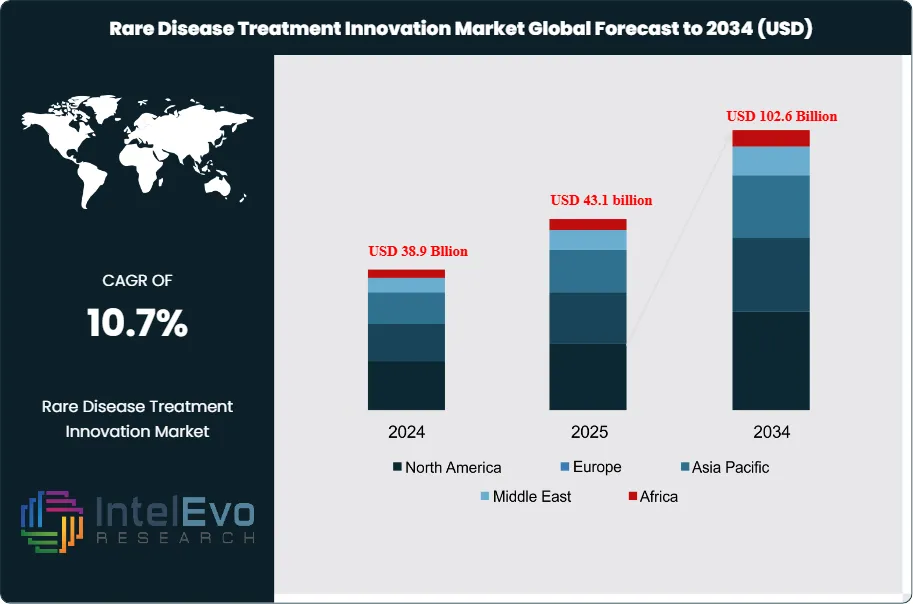
The Global Rare Disease Treatment Innovation Market size is expected to be worth around USD 102.6 billion by 2034, up from approximately USD 38.9 billion in 2024, growing at a CAGR of 10.7% during the forecast period from 2025 to 2034. With advancements in gene therapy, precision medicine, and AI-driven drug discovery, the rare disease treatment ecosystem is witnessing unprecedented growth. Increasing global healthcare investments and strong regulatory support are further driving innovations that bring hope to millions of patients worldwide. This sector is positioned to reshape the future of healthcare with breakthrough treatments and collaborative R&D efforts.
The Global Rare Disease Treatment Innovation Market encompasses therapies developed for the treatment of rare diseases, which are conditions affecting a small percentage of the population. The market is characterized by a range of innovative products, including biologics and non-biologics, aimed at addressing unmet medical needs. Current dynamics indicate a growing interest in rare diseases, driven by advancements in research and development, increasing drug approvals, and a robust pipeline from major pharmaceutical companies. Regulatory incentives and the globalization of healthcare further contribute to the market's expansion, reflecting a commitment to improving patient outcomes and access to treatments for these challenging conditions.
The growth dynamics of the rare disease treatment market are underscored by several key drivers. Increasing awareness of rare diseases among healthcare providers and patients is propelling demand for effective treatments. Advances in gene therapy, precision medicine, and orphan drug development have expanded the therapeutic landscape, enabling new treatment modalities that were previously unavailable. Additionally, government support and funding for research initiatives are fostering innovation in this sector. The rising prevalence of rare diseases and the associated push for personalized medicine are also critical factors contributing to the market's growth. These elements collectively create a favorable environment for investment and development in rare disease treatments.
North America is anticipated to dominate the rare disease treatment market, attributed to high healthcare spending, advanced research facilities, and a favorable regulatory environment. The presence of key market players and a robust pipeline of therapies further reinforce this dominance. However, the Asia-Pacific region is expected to witness significant growth, driven by increasing healthcare investments, rising awareness, and government initiatives to improve access to treatments. Europe also plays a crucial role in the market, supported by strong healthcare systems and a focus on rare disease research and development. As global healthcare systems evolve, the market landscape will continue to change, highlighting the importance of regional strategies.
The COVID-19 pandemic has had a notable impact on the rare disease treatment market, influencing research, development timelines, and patient access to therapies. Disruptions in clinical trials and healthcare services affected the development and availability of new treatments, leading to delays in approvals. However, the pandemic also accelerated the adoption of telemedicine and remote patient monitoring, which may benefit the treatment of rare diseases in the long run. As healthcare systems adapt to the new normal, the focus on rare diseases is expected to strengthen, ensuring that patients receive the necessary care and attention.
Get More Information about this report -
Request Free Sample Report
Key Takeaways:
- Market Growth: The global rare disease treatment innovation market is expected to reach USD 102.6 billion by 2034, growing at a robust CAGR of 10.7%, indicating strong market expansion due to advancements in research and increased awareness.
- Disease Type Analysis: The market is predominantly segmented by disease type, with cancers, metabolic diseases, and neurological conditions accounting for significant shares. The rising prevalence of these conditions drives demand for innovative therapies targeting specific rare diseases.
- Product Type Analysis: The product type segment is primarily led by biologics, reflecting their effectiveness in treating complex rare diseases. The increasing approval of biologics by regulatory agencies supports market growth and offers diverse treatment options for patients.
- Driver: Key growth drivers include heightened awareness of rare diseases, advancements in gene therapy, and supportive government policies promoting orphan drug development, leading to a favorable environment for innovation and investment in this sector.
- Restraint: High costs associated with the development and production of rare disease treatments pose a significant restraint. Limited patient populations for rare diseases can also hinder the financial viability of developing new therapies.
- Opportunity: The growing focus on precision medicine and personalized therapies presents substantial opportunities for market expansion. Additionally, increasing investments in rare disease research from pharmaceutical companies can lead to breakthrough treatments.
- Trend: A significant trend in the market is the rising adoption of gene therapy and personalized medicine, enabling tailored treatments that address the specific genetic causes of rare diseases.
- Regional Analysis: North America is expected to dominate the market, driven by high healthcare expenditures and advanced research capabilities, while the Asia-Pacific region shows promising growth potential due to increasing healthcare investments and awareness initiatives.
Disease Type:
The disease type segment of the global rare disease treatment market includes a variety of conditions, with significant contributions from cancer, metabolic diseases, and neurological disorders. Cancers, particularly rare subtypes, account for a large portion of the market due to the urgent need for targeted therapies. Metabolic diseases, such as phenylketonuria (PKU) and Gaucher disease, drive demand for innovative treatments, highlighting the importance of enzyme replacement and gene therapy. Neurological conditions, like Huntington's disease and spinal muscular atrophy, are also key contributors, prompting research into advanced therapeutic options. The growth in this segment is bolstered by increasing awareness and diagnosis, coupled with advancements in medical research and a focus on developing orphan drugs for these underserved conditions.
Product Type:
The product type segment is primarily categorized into biologics and non-biologics, with biologics leading the market. Biologics, which include monoclonal antibodies, gene therapies, and cell-based therapies, have transformed treatment paradigms for various rare diseases. Their ability to target specific pathways and provide personalized treatment options has garnered significant attention from healthcare providers and patients alike. Non-biologics, encompassing traditional small molecule drugs, also play a vital role in treating rare diseases but face challenges related to efficacy and safety compared to biologics. The increasing number of biologics receiving regulatory approval is fueling market growth, reflecting a trend towards innovative and targeted therapies that offer improved outcomes for patients suffering from rare conditions.
End-User:
The end-user segment of the rare disease treatment market is primarily composed of specialty pharmacies, hospital pharmacies, and retail pharmacies. Specialty pharmacies are pivotal in managing the distribution of complex and high-cost therapies, particularly biologics and advanced therapeutics, ensuring patients receive proper education and support. Hospital pharmacies are also crucial, as they provide on-site dispensing and clinical management for patients undergoing treatment in healthcare facilities. Retail pharmacies play a complementary role, offering access to non-specialty drugs and over-the-counter treatments for rare diseases. The growing prevalence of rare diseases and the increasing complexity of treatments necessitate specialized services, driving growth within specialty and hospital pharmacies as they adapt to meet the evolving needs of patients and healthcare providers.
Age Group:
The age group segment is divided into adult and pediatric categories, with distinct needs and treatment approaches for each group. Pediatric patients often require specialized formulations and dosing, as their physiological differences necessitate tailored therapies. This segment has seen a rise in the development of treatments targeting genetic disorders, congenital conditions, and metabolic diseases prevalent in children. Adult patients, on the other hand, are more frequently diagnosed with conditions like rare cancers and autoimmune diseases. The market's growth in both segments is driven by increased awareness, advancements in genetic testing, and a greater focus on developing innovative therapies that address the specific challenges faced by different age groups. The expansion of research initiatives targeting both pediatric and adult rare diseases continues to shape this segment.
Region Analysis:
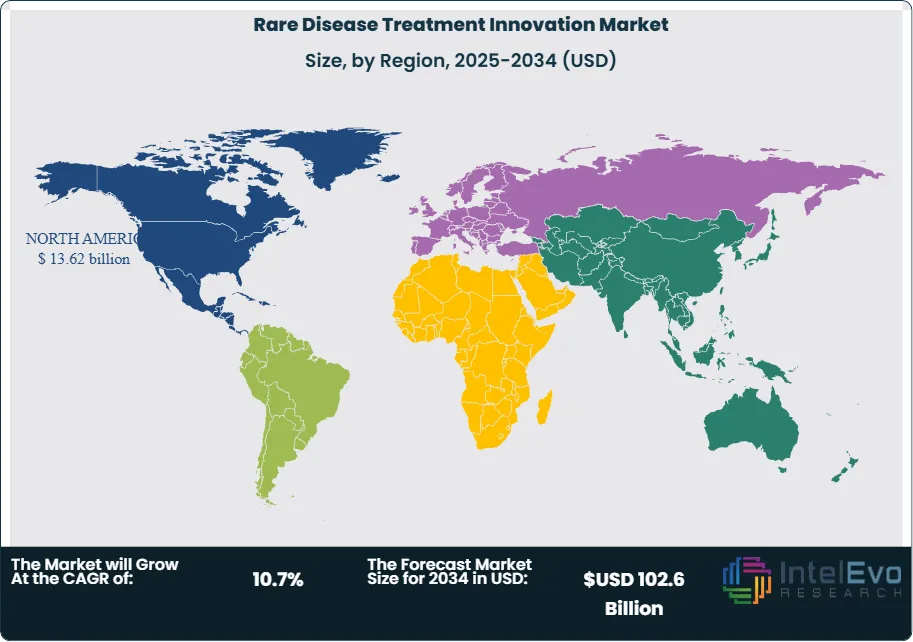
North America Leads With 35% Market Share in the Global Rare Disease Treatment Innovation Market: North America holds the largest market share in the global rare disease treatment innovation market, accounting for approximately 35% of the total revenue. This dominance is primarily driven by advanced healthcare infrastructure, significant investments in research and development, and a high prevalence of rare diseases. The United States, in particular, has a robust ecosystem of pharmaceutical companies and research institutions focused on developing orphan drugs and biologics. Furthermore, supportive government policies, such as the Orphan Drug Act, incentivize the development of therapies for rare conditions. The presence of well-established healthcare systems, alongside increasing patient awareness and advocacy, fuels market growth. Additionally, the growing trend of personalized medicine and gene therapy is anticipated to strengthen North America's position in the market further.
The Asia-Pacific region is emerging as the fastest-growing market for rare disease treatment innovation, with a projected CAGR of over 12% during the forecast period. Key drivers include increasing healthcare investments, expanding pharmaceutical markets, and rising awareness of rare diseases among healthcare professionals and patients. Countries like China and India are making substantial advancements in biotechnology and research capabilities, leading to the development of innovative therapies tailored to local populations. Additionally, supportive government initiatives and funding for rare disease research are fostering growth in this region. Meanwhile, Europe remains a strong player, accounting for a substantial market share, driven by a well-established healthcare system and comprehensive regulations supporting orphan drug development. Latin America and the Middle East and Africa also exhibit growth potential, albeit at a slower pace, due to improving healthcare access and rising incidences of rare diseases.
Get More Information about this report -
Request Free Sample ReportKey Market Segment
By Type
- Biologics
- Non-Biologics
By Treatment Type
- Gene Therapy
- Enzyme Replacement Therapy (ERT)
- Small Molecule Drugs
- Monoclonal Antibodies
- Cell Therapy
- Others (Supportive & Symptomatic Therapies)
By Therapy Type
- Disease-Modifying Therapies
- Symptomatic Treatments
By Route of Administration
- Oral
- Injectable
- Others
By Age Group
- Adult
- Pediatric
By Distribution Channel
- Hospital Pharmacies
- Specialty Clinics
- Retail Pharmacies
- Online Pharmacies
By Application (Disease Area)
- Oncology Rare Diseases
- Neurological Rare Diseases
- Hematological Rare Diseases
- Metabolic & Genetic Disorders
- Others
By End-User
- Hospitals & Clinics
- Research Institutes
- Specialty Care Centers
- Others
By Region
- North America
- Latin America
- East Asia And Pacific
- Sea And South Asia
- Eastern Europe
- Western Europe
- Middle East & Africa
| Report Attribute | Details |
| Market size (2025) | USD 43.1 billion |
| Forecast Revenue (2034) | USD 102.6 Billion |
| CAGR (2025-2034) | 10.7% |
| Historical data | 2018-2023 |
| Base Year For Estimation | 2024 |
| Forecast Period | 2025-2034 |
| Report coverage | Revenue Forecast, Competitive Landscape, Market Dynamics, Growth Factors, Trends and Recent Developments |
| Segments covered | Type (Biologics, Non-Biologics), By Treatment Type (Gene Therapy, Enzyme Replacement Therapy (ERT), Small Molecule Drugs, Monoclonal Antibodies, Cell Therapy, Others (Supportive & Symptomatic Therapies)),By Therapy Type (Disease-Modifying Therapies, Symptomatic Treatments), By Route of Administration (Oral, Injectable, Others), Age Group (Adult, Pediatric), By Distribution Channel (Hospital Pharmacies, Specialty Clinics, Retail Pharmacies, Online Pharmacies), By Application (Disease Area), Oncology Rare Diseases, Neurological Rare Diseases, Hematological Rare Diseases, Metabolic & Genetic Disorders, Others), 8 By End-User (Hospitals & Clinics, Research Institutes, Specialty Care Centers, Others) |
| Research Methodology |
|
| Regional scope |
|
| Competitive Landscape | Roche, Novartis, Sanofi, GSK (GlaxoSmithKline), Pfizer, Amgen, Biogen, Vertex Pharmaceuticals, Alexion Pharmaceuticals, Eli Lilly and Company, Bristol Myers Squibb, Merck & Co., AbbVie, Regeneron Pharmaceuticals, Sarepta Therapeutics, Orchard Therapeutics, Bluebird Bio, Ultragenyx Pharmaceutical, Takeda Pharmaceuticals, AstraZeneca |
| Customization Scope | Customization for segments, region/country-level will be provided. Moreover, additional customization can be done based on the requirements. |
| Pricing and Purchase Options | Avail customized purchase options to meet your exact research needs. We have three licenses to opt for: Single User License, Multi-User License (Up to 5 Users), Corporate Use License (Unlimited User and Printable PDF). |
Select Licence Type
Connect with our sales team
Rare Disease Treatment Innovation Market
Published Date : 24 Dec 2024 | Formats :Why IntelEvoResearch
100%
Customer
Satisfaction
24x7+
Availability - we are always
there when you need us
200+
Fortune 50 Companies trust
IntelEvoResearch
80%
of our reports are exclusive
and first in the industry
100%
more data
and analysis
1000+
reports published
till date







